A Medical Device Contract Manufacturing Organization is never at rest. With diversity of projects from different segments of Medical Device Industry, the possibilities of errors, risks, and upgrades are infinite. Every new project comes with its challenges and opportunities to learn. The learnings gained over the years help in refining the processes and staying in sync with latest requirements of the Medical Device Industry.
With cascaded processes involved in Medical Device Development some errors may get unnoticed over a period of time. These errors might look small initially, but when left unattended for a long these may lead to bigger problems. Analysis of these issues and measures adopted to eliminate them is tagged under the banner of “CAPA”. CAPA (Corrective Action Preventive Action) refers to a set of practices adopted to correct an error that occur repetitively.
3 Main Elements of a CAPA (Corrective Action Preventive Action)
1.A continually evolving development system
Every time we implement CAPA the aim is not to solve a single issue. Rather, it is to continually eliminate the cumulative errors and make the system more robust and productive.
2.Defined KPIs for continual monitoring & analysis
The next step is to set & evaluate proper key performance indicators. The Lean Six Sigma based manufacturing facilities believe in keeping the system Lean by optimizing the human resource too. A regular overview and mapping of the resource performance is necessary to identify priorities and eliminate or optimize unused resources.
3. Outcome oriented functioning
A CAPA should be quantifiable and must have clear metrics to evaluate the success of the entire activity.
How to implement a CAPA plan in Medical Device Manufacturing?

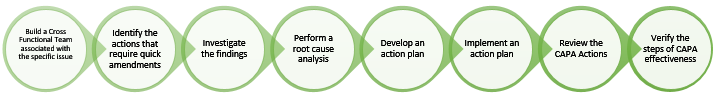
Steps post CAPA Request Acceptance
- Build a Cross Functional Team with members involved from all the departments in manufacturing facility
- Identify the actions that require quick amendments
- Investigate the findings
- Perform a root cause analysis
- Develop an action plan
- Implement an action plan
- Review the CAPA Actions
- Verify the steps of CAPA effectiveness
When should you plan to implement CAPA in a medical device manufacturing organization?
In the Medical Device Industry one can implement CAPA in these 6 key areas.

1.) In case of Part Failure
In cases where a Part or Component in a Medical Device causes severe problems, a CAPA can be initiated to identify the root cause and implement changes.
2.) In case of Documentation Error
Documentation is an important part of Medical Device Development. A minor glitch in documentation (DHR, DMR, DHF) can have serious repercussions. Therefore, in cases when any documentation is not right, a CAPA is necessary.
3.) In case of Process Error
Training might get outdated; Machinery might need an upgrade or it can be anything. Repetitive errors can be a process defect too. Often, when error has a significant mass impact, it is important to trickle down to the basics of every process. A CAPA is needed to identify & eliminate the process defect.
4.) In case of Operations Error
Issues in Inventory Management, Supply Chain Issues, Supplier or vendor issues, a CAPA is necessary to improve the processes and identify the root cause of the problem.
What is the best time to implement a CAPA?
The QA/RA teams must know exact situations when a CAPA is needed. A CAPA would require involvement from many departments which means it will open a new thread which needs a lot of rolling. It’s necessary to understand the gravity of the issue for which the CAPA is being implemented.
CAPA is only required when an issue is absolutely pressing and has a very vast impact. A small issue which has minor or no impact doesn’t need a lot of attention and time. Prioritization is a must to keep things rolling without any major hurdles or obstacles.
Conclusion
At Johari, we ensure 100% risk analysis based Medical Device Development process. At every step of manufacturing, we implement stringent controls to minimize errors and maximize productivity & timelines. Are you looking for a reliable Contract Medical Device Manufacturing partner to assist you in fast Go-To-Market. Get in touch with us to explore dependable, Regulatory Compliant & Cost optimized Medical Device Contract Manufacturing Services.


Hello . I register a medical device. which is part of orthopedics and another part is the possibility of treating and relieving pain. It is used for children and adults. And due to its important nature and ease of use, it is used by important sections of the society both for treatment and prevention, as well as for exercise and growth. I am looking for its production and investment. Do you work in this field?